Speed to Market: How Gen AI accelerates MLR Reviews for Pharma
Pharmaceutical marketing has evolved significantly with digital platforms, but strict regulations ensure patient safety at every stage. Compliance is crucial errors can result in costly penalties or halted production. The Medical, Legal, and Regulatory (MLR) review process plays a key role in ensuring promotional content adheres to guidelines while delivering accurate medical information.
However, challenges such as inconsistent reviews, evolving regulations, redundant content creation, and version-tracking issues often slow down drug launches. Our Gen AI-powered MLR solutions address these inefficiencies by automating compliance verification and content adaptation, enabling faster and more accurate reviews.
The growing complexity of Pharma advertising
Pharmaceutical promotional practices are crucial for launching new drugs and ensuring they reach the right patients and healthcare providers. However, every piece of advertising content must first pass through the Medical, Legal, and Regulatory (MLR) review process to ensure safe, accurate drug details get promoted. Despite this, innovation in the pharma industry has not always translated into ethical marketing, with some companies resorting to questionable tactics, leading to financial and legal repercussions.
As marketing trends evolve, pharmaceutical companies continuously expand their product portfolios and campaigns. However, with regulatory bodies worldwide frequently updating drug marketing guidelines, it becomes challenging to ensure complete compliance. Failing to adhere to these regulations results in non-compliant advertisements, which not only affect patient safety but also invite hefty penalties.
To address these challenges, LTIMindtree has developed two AI in Pharma Compliance solutions powered by generative AI. These solutions assist content planners in redesigning advertisements to meet updated marketing regulations across different platforms (print/digital) and support MLR reviewers by analyzing promotional content and generating compliance reports, significantly improving the review process.
Generative AI solutions for MLR compliance
Generative AI streamlines the MLR review process by automating repetitive tasks, reducing manual effort, and ensuring data security. With advanced technologies, such as Large Language Models (LLMs), Small Language Models (SLMs), Natural Language Processes (NLPs), and Retrieval Augmented Generations (RAGs), Gen AI can optimize MLR compliance in multiple ways.
MLR workflow process presentation
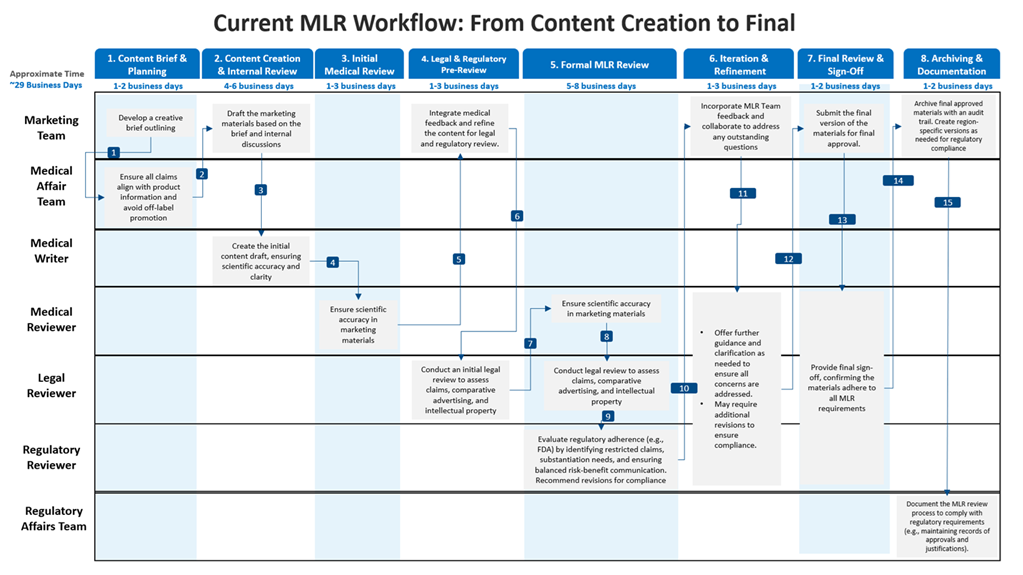
Fig 1: The Medical, Legal, and Regulatory review process of one marketing material takes around twenty-nine business days to get commercialized
LTIMindtree’s MLR review assist
Pharmaceutical drug marketing guidelines vary in different countries. Therefore, if a drug needs to be launched in a country, the manufacturing company must follow the respective regulatory drug marketing guidelines (CDSCO, USFDA, TGA, MHRA, EMA, etc.)
Challenges- Inconsistent review processes delay drug launches
- Marketing materials often fail to keep up with evolving regulatory guidelines
- Repetitive and inefficient reviews by subject-matter experts
- Content redesign efforts increase due to platform-specific advertising formats
Gen AI-powered solution and business benefits
- Analyzing drug advertisements for compliance
- LTIMindtree’s MLR Review Assist, built with LLMs, is trained on company branding guidelines (logo, font style, color theme, etc.), regulatory standards (FTC, USFDA, etc.), and the drug’s clinical data.
- It accelerates the identification of discrepancies between advertising claims, clinical data, and regulatory standards, helping MLR reviewers expedite the process.
- Identifying non-compliance and generating compliance score report
- MLR Review Assist categorizes advertisement content into “Compliant” and “Non-Compliant” under four key areas: Medical, Legal, Regulatory, and Company Branding Guidelines. This helps generate a compliance score report in percentage and identify non-compliance data in advertisements.
- This prevents MLR reviewers from recurrent review cycles and allows them to suggest their expertise in a focused domain, leading to a consistent, time-efficient review process.
- Providing attribution to reference documents
- The tool highlights the reference data points used to classify compliance and non-compliance claims.
- This allows MLR reviewers and content planners to validate the compliance score and identify the root cause of non-compliance efficiently.
Recreating marketing content with LTIMindtree’s MLR content bot
Marketing teams often repurpose content across multiple platforms, making it a repetitive and time-consuming task. LTIMindtree’s MLR Content Bot automates content creation while maintaining accuracy and compliance.
Challenges
- Repetitive task in commercial creation: Advertising single medical product commercials in different marketing platforms with consistent accurate details is a repetitive, non-creative task for marketing content creators
- Lack of personalization in marketing materials
- Limited reach due to language barriers
Generative AI solutions and business benefits
- Transforming marketing content into emails and social media posts
- The MLR Content Bot, powered by image-to-text LLMs, reformats advertisements into professional email bodies and engaging social media posts.
- It ensures compliance while maintaining an engaging tone with relevant hashtags and taglines.
- Language translation for global reach
- The bot translates marketing content into multiple languages, including Spanish and German.
- This expands the reach of innovation in the pharma industry, making drug information accessible to a wider audience.
- Persona-based advertising
- The bot customizes marketing content based on demographics such as age, gender, and patient category (e.g., pregnant women, elderly, children).
- This ensures that drug promotions are tailored for different consumer segments, enhancing engagement and compliance.
Future of Gen AI in MLR compliance
Generative AI can enhance the MLR process with its ability to generate analyzed reports with evidence-based data sources. In the upcoming years, Gen AI can accelerate most of the time-consuming processes of pharmaceutical marketing with the following add-on specifications:
- Automating regulatory submission document creation.
- Enabling personalized patient communication through AI-driven messaging.
- Supporting legal teams by summarizing and reviewing compliance-related legal documents.
- Real-time tracking of reviewed marketing content to ensure ongoing compliance.
Conclusion
Generative AI is reshaping the innovation in the pharma industry, making the MLR review process more efficient, accurate, and compliant. By addressing challenges in review time and content creation, AI-powered solutions offer a faster, smarter, and more precise approach to pharmaceutical marketing compliance. As AI in pharma industry continues to advance, it will further enhance compliance, accelerate drug approvals, and improve patient engagement.
Latest Blogs
A closer look at Kimi K2 Thinking, an open agentic model that pushes autonomy, security, and…
We live in an era where data drives every strategic shift, fuels every decision, and informs…
The Evolution of Third-Party Risk: When Trust Meets Technology Not long ago, third-party risk…
Today, media and entertainment are changing quickly. The combination of artificial intelligence,…




